|
At this point oxidation reaction take place and negative ions or anions are attracted and lose an electron to become neutral atoms or molecules. It is the type of electrode that is a negative charge in an electrochemical system. 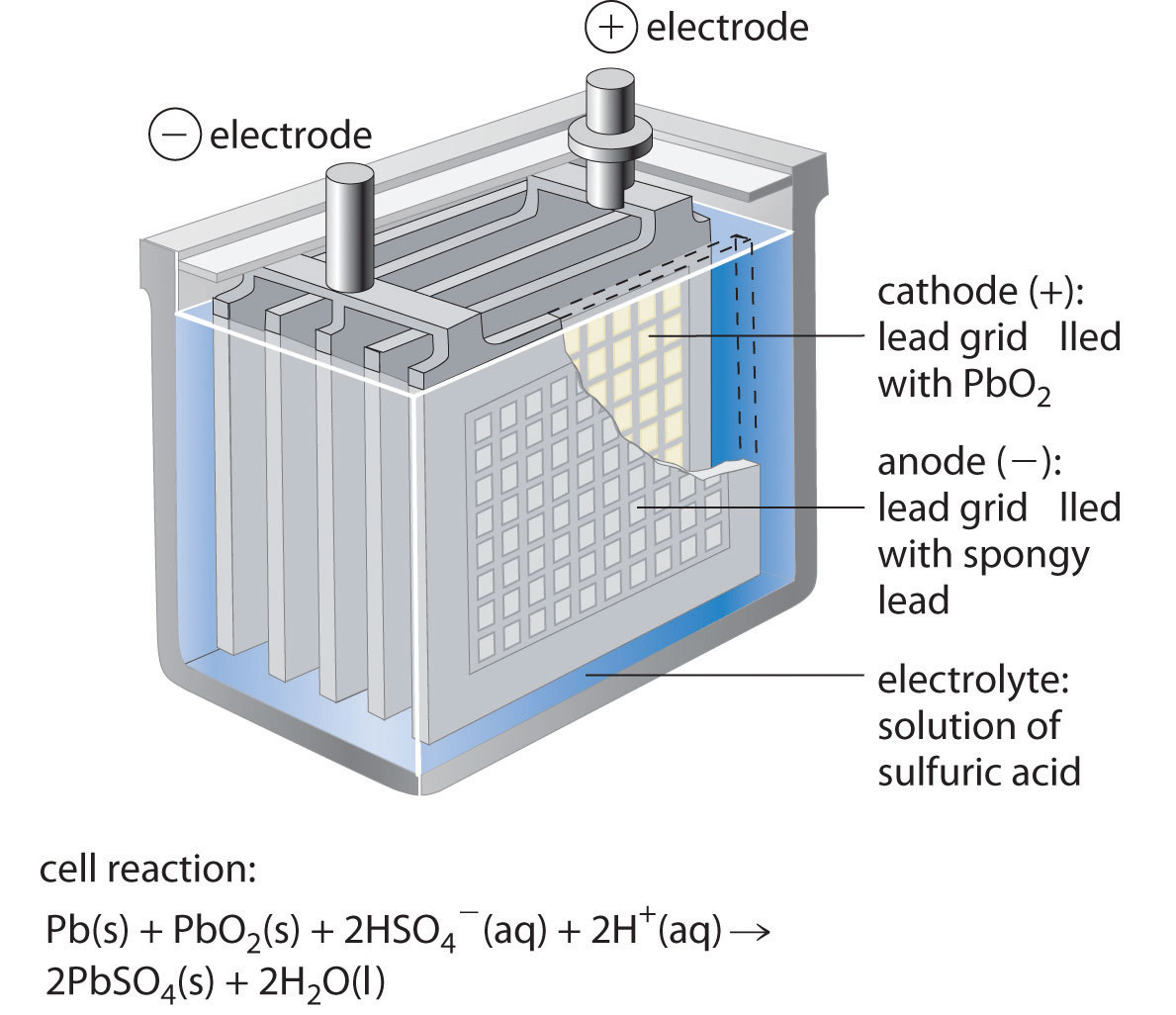
Positive electrodes are used in different applications such as batteries, electrolysis cells, and fuel cell, in these applications they help to generate and store electrical energy.In the case of a cell or battery positive electrodes are called the cathode and it is attached to the positive terminal of the power source.It is considered as the site of reduction reaction since positive charges or cations are moved to the electrode and get electrons and make neutral atoms or molecules. The positive electrode is a type of electrode that has a positive charge.Functions: Electrons are produced at the anode and cathodes get the electrons.Polarity: The anode is attached to a positive point of the battery or other power sources and the cathode exists at the negative terminal of the battery.Cathode has a reduction reaction and exists at the right side Position in a cell: In the case of a galvanic cell the electrode where oxidation occurs is called the anode and exists on the left side.Chemical Reaction: Oxidation take place at the anode and loss of electrons occurs while at the cathode electrons gain and a reduction reaction occurs.Electron flow direction: The direction is anode to the cathode for electrochemical cells while for diodes and other electronic devices the direction of current from anode to cathode.Charge: The cathode is negatively charged and the anode has a positive charge.Here are some points discussed for comparison.: 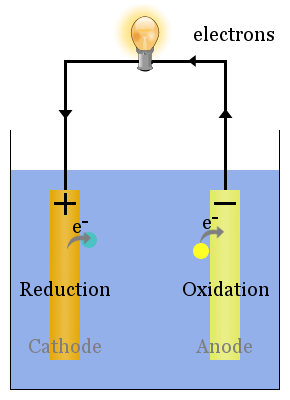
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
